The oceans are now acidifying faster than they have been over the last 300 million years, a time period in which there were four major extinctions driven by natural bursts of carbon. In fact, humans are acidifying the oceans 10 times faster today than 55 million years ago when a mass extinction of marine species occurred.
Recent research finds that the threat to marine life posed by human-caused carbon pollution is coming faster than expected. And that’s a problem because as 70 Academies of Science warned in a 2009 joint statement on acidification: “Marine food supplies are likely to be reduced with significant implications for food production and security in regions dependent on fish protein, and human health and wellbeing.”
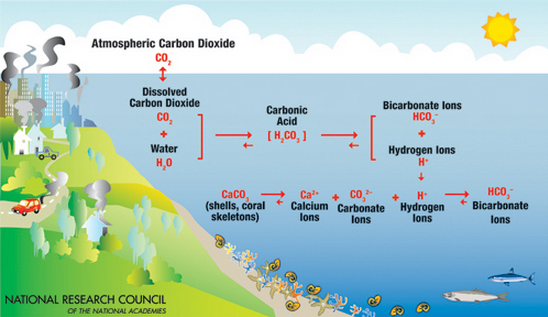
Why does carbon pollution threaten marine life? Significantly, as carbon dioxide is absorbed in water it causes chemical reactions that reduce “saturation states of biologically important calcium carbonate [CaCO3] minerals,” which “are the building blocks for the skeletons and shells of many marine organisms,” as NOAA explains. In the parts of the ocean teeming with life, the seawater has an overabundance (supersaturation) of these calcium carbonate minerals used by so-called “calcifying organisms,” which include corals and algae and mollusks and some plankton.
As the ocean absorbs more carbon dioxide, more and more places are becoming undersaturated with these minerals, thereby threatening calcifying organisms. Besides a decline in calcification, the World Meteorological Organization explained in 2014, “Other impacts of acidification on marine biota include reduced survival, development and growth rates, as well as changes in physiological functions and reduced biodiversity.”
A 2015 study in Science concluded that the Permo-Triassic extinction 252 million years ago — considered the “the greatest extinction of all time” — happened during the time when massive amounts carbon dioxide were injected into the atmosphere, first slowly and then quickly (driven by volcanic eruptions). The researchers found “During the second extinction pulse, however, a rapid and large injection of carbon caused an abrupt acidification event that drove the preferential loss of heavily calcified marine biota.” How bad was this extinction? Besides killing over 90 percent of marine life, it wiped out some 70 percent of land-based animal and plant life.
Today, coral reefs alone are estimated to support a quarter of all marine life. NOAA explains that “The fish that grow and live on coral reefs are a significant food source for half a billion people worldwide.” The combination of warming waters and acidification have already caused serious harm to major coral reef around the world, and many appear unlikely to survive the century. Oceanographer and coral expert J.E.N. Veron, former chief scientist of the Australian Institute of Marine Science, has written, “The science is clear: Unless we change the way we live, the Earth’s coral reefs will be utterly destroyed within our children’s lifetimes.”
Ocean acidification and carbon pollution have already proven to be major threats to the U.S. oyster industry, as was clear from the “The Great Oyster Crash” of 2007 in coastal Oregon and Washington. There were “near total failures of developing oysters in both aquaculture facilities and natural ecosystems on the West Coast,” as NOAA put it, with oyster larvae dying by the millions. Why? Originally it was thought that rapidly acidifying coastal waters made it difficult for larvae to build the shells needed for survival.
But a December 2014 study of Pacific oyster and Mediterranean mussel larvae in Nature Climate Change determined that “the earliest larval stages are directly sensitive to saturation state, not carbon dioxide (CO2) or pH” (acidity). So what matters most is how much calcium carbonate is in the ocean water relative to the total amount it could hold.
This finding has dramatic consequences for the speed at which rising carbon dioxide levels will harm ocean life. Lead author George Waldbusser, an Oregon State University marine ecologist and biogeochemist explains why:
Larval oysters and mussels are so sensitive to the saturation state (which is lowered by increasing CO2) that the threshold for danger will be crossed “decades to centuries” ahead of when CO2 increases (and pH decreases) alone would pose a threat to these bivalve larvae. “At the current rate of change, there is not much more room for the waters off the Oregon coast to absorb more CO2 without crossing the threshold we have identified with respect to saturation state,” he said.
That means some of the worst impacts of rising carbon dioxide levels in the ocean may come sooner than expected.
